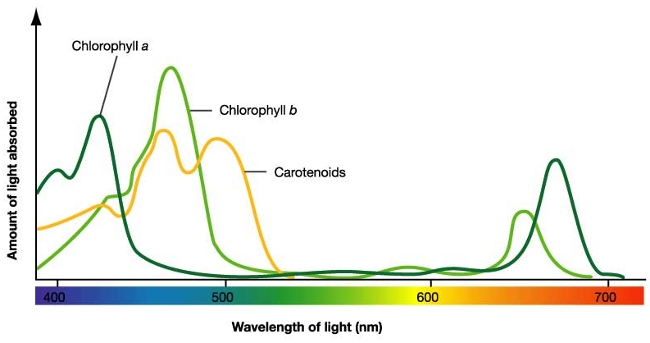
| Measurement of what? | Name of unit | Symbol | Note(s) |
|---|
| length (distance) | meter | m | one ten millionth (10-7) of the distance from the North Pole to the equator on a line (the meridian) passing through Paris |
| mass | kilogram | kg | mass of a certain reference at the International Bureau of Weights and Measures |
| time | second | s | no short answer but probably originally of Babylonian derivation (they counted in 60s) but re-defined in terms of an atomic frequency |
| electric current | ampere | A | strange circular definition of this and
the coulomb (C; unit of charge) 1 A = 1C per s. |
| temperature | kelvin | K | The size of the unit is the same as the Celsius scale
i.e. 1 degree K = 1 degree C but whereas 0 C is the temperature of melting ice, 0 K is the lowest temperature
that is absolutely possible "absolute zero"; 0 C = 273.15 K |
| chemical amount | mole | mol | "gram molecule". Note this is not
an abstract idea: 1 mol of water is 18 grams. |
| luminous intensity | candela | cd | I think an arbitrary unit |