Self-assessment questions
The questions all follow the same convention: questions in
this font can be answered from the text of the
pages of the topics in the module and are potentially useful in
helping you to
decide whether you need to study the topic at all. Questions in
dark green range slightly
outside the confines of the topic but the answers should lie within
the module as a whole. Questions in
red require some work in
libraries or on the Web.
-
Biological chemistry in time and space
-
Molecules: structures, isomers, chirality, representations of
molecular structures
- Equilibrium and reactions
-
The biological and chemical literature
-
Macromolecules; primary structures and conformations
-
Genomes, gene expression and protein biosynthesis
-
Metabolic, regulatory and neural networks
-
Classification and ontologies in the biological sciences
-
Recombination, repair, rearrangement and evolution
TOP of page
Topic 1
- Draw a straight line representing the "history of
everything" from the big bang to the present day.
- what is the approximate length of the line in years?
- mark the approximate position of the supposed origin of life on
earth
- Mark on the line approximately the positions corresponding to
origins of the classes Oo4 - Oo7 of Chandler's
semiotics
- Within an order of magnitude what are the sizes in mm
(millimetres) of an oxygen molecule (O2),
a small globular protein, a small virus,
a bacterium?
- Define (or give examples) of the word, "ecoment";
differentiate between a biolgist's use of the word, "cell"
and the class Oo5 of Chandler: speculate
wildly (but briefly!) on why Chandler might have eschewed the
biologist's definition.
- Give one example each of the importance in biochemistry of
interactions between a macromolecule and (a) a smaller molecule and
(b) another macromolecule.
TOP of page
Topic 2
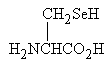
- What is the valency of Se?...
... and what is the significance in biochemistry of the molecule shown below?
 What is the shape of each of the following?
What is the shape of each of the following?
- an ammonium ion (NH4+)
- ethylene (H2C=CH2)
- H2C=C=CH2 (it doesn't matter what it's
called)
- an oxonium ion (H3O+)
What does SMILES stand for? and
what are the SMILES strings for methane, ethane, benzene, phenol
and D-glyceraldehyde?
Define (or explain briefly with examples) enantiomer, geometric
isomer, diastereoisomer.
Complete the following table (C6H5- is
phenyl)
| Rough formula |
types of isomerism | acidic, basic? | reducing/oxidising |
| CH3CH2OH |
| | |
|
C6H5-OH |
| | |
| CH3CH2NH2 |
| | |
| CH3CH=O |
| | |
| CH3C(=O)OH |
| | |
| HO(O=)C-CH=CH-C(=O)OH |
| | |
| (CH=O)-CHOH-CH2OH |
| | |
|
(CH2OH)2C=O
|
| | |
Which of the molecules in the pictures below contain chiral C atoms?
 What is the relationship between CH3CH=O
("acetaldehyde") and CH2=CH2OH
("vinyl alcohol")?
Write down the products if each of the following
is hydrolysed.
What is the relationship between CH3CH=O
("acetaldehyde") and CH2=CH2OH
("vinyl alcohol")?
Write down the products if each of the following
is hydrolysed.
- CH3(C=O)NHCH3
- CH3(C=O)OCH3
- (HO)2P(=O)OCH3
-
CH3CH(OCH3)2
TOP of page
Topic 3
Note: questions in bold are important.
- What is the molarity (M) of pure water?
- Glycerol is CH2OH-CHOH-CH2OH
- What is the RMM of glycerol?
-
What is the mass of glycerol (in g) in 250 ml of a 150 mM
glycerol solution?
- So far we have looked at rather small molecules. What would
be the mass of 1 mol of a large protein of RMM 105?
- Look at the
acetic acid/acetate saga. Note
that if we add a strong acid to such a buffer, we substract the
concentration of strong acid from [OAc-] and add the value
to [HOAc] and likewise with a strong base we add the value to
[OAc-] and subtract it from [HOAc]. Assume the pK is
4.0. We have a solution of 100 mM HOAc and 120 mM NaOAc in our
buffer.
What are the effects of adding ....
- HCl to a concentration of 1mM to water
- HCl to a concentration of 1mM to our buffer
- NaOH to a concentration of 1mM to water
- NaOH to a concentration of 1mM to our buffer ?
- Re-write the text of the account of
buffers using as an example a solution
of NH3 and NH4Cl.
Specifically, what is the
form of the equation
"pH = pK + log10(something-or-other)" in this
case?
- Rate constants etc.:
- What is the equation ("kt = ln(something)")
for the bimolecular reaction of
A and B?
- How would the answer to that differ if
a and b were equal?
- Define or explain briefly, free energy, free energy change,
standard free energy change.
What is the free energy change for a reaction at equilibrium and
how does this differ from the free energy change for a reaction
not at equilibrium?
- Write (hopefully short !) essays on
- transition states
-
the significance
of the parameter A in the Arrhenius equation
TOP of page
Topic 4
These are exercises in using the Web.... do not infer that
that the answers themselves are crucial to understanding
bioinformatics. Following our rules, they should all be
in red.
- What type of sea creature contains vanadate that replaces, to some
extent, phosphate in animals such as ourselves?
- Name one human protein that contains copper ions.
- Find a tutorial on chemcial reaction rates.
- What is the geographical, as opposed to biological, significance
of the word "wombat"?
- Where is the largest taxonomic resource in Belgium?
- What are the modern terms for the following?
- muriatic acid
- spirits of salt
- old yellow enzyme
- new yellow enzyme
- Zwischenferment
-
What is the full and correct chemical name for the substance
biochemists call GTP?
- What are the chemical components of fusel oil?
- What is "fuller's earth" and why is not referred to as
"Fuller's earth"?
- What is the importance of Streptomyces rimosus in the
pharmaceutical industry?
TOP of page
Topic 5
- What are differences (if any) between
- a peptide bond and an amide bond
- an O-glycoside and an N-glycoside
- α and β anomers
- a nucleotide and a nucleoside
- Name one or more amino acid(s) that are in some sense similar
to
- methioine
- glutamic acid
- tryptophan
- name or describe a phosphomonoester and a phosphodiester
- Print out the following pictures and
draw an approximate
scale (Å):
- Now do question 4 again using the
correct sizes
- Have a look at RasMol. It can be obtained from
www.umass.edu/microbio/rasmol/getras.htm and elsewhere (try Google).
- Here is a DNA sequence 5'-to-3' left-to-right
ACATAAACATGGGACCCAA
Write down the sequence of the complementary strand
also 5'-to-3' left-to-right.
TOP of page
Topic 6
- Explain the terms diploid and allele
- What is a ribozyme?
- In what forms of life would you expect to find ...?
- introns
- polycistronic mRNA
- Explain the concept of "template" in gene expression
- Describe qualitatively (e.g. pseudocode) a method for looking
up the Genetic Code in a computer program.
- Explain what is meant by "reading frame".
- In our example of reading frames
one of us (!)
felt confident in writing down 10 amino acids in the reading frames
labelled "2" but only felt able to write donw 9 in the
case of reading frames "3". Why was this?
TOP of page
Topic 7
- Look at the description of
Michaelis-Menten kinetics
- A competitive inhibitor has the effect of increasing
KM: sketch the graph for this hypothetical
enzyme in the presence of such an inhibitor
- What is the value of [S] when v =
Vmax/2 ?
- Vmax and KM can be calculated by
doing a non-linear regression to fit v versus [S] data to the
equation. In the era before everday use of computers, biochemists
used to plot 1/v as a function of 1/[S] to obtain a straight line
that could be fitted by hand.
- Sketch such a graph and label
Vmax and KM
- However this is
an inaccurate method: why?
- Discuss briefly with examples anhydrides and esters of
phosphoric acid.
- Name or describe one reaction or process in which a
favourable overall free energy change is achieved by the hydrolysis
of GTP rather than ATP and speculate on how the resultant GDP
might be re-converted to ATP.
- In an important metabolic reaction, glucose-6-phosphate is
oxidised in a REDOX reaction
-
What is glucose-6-phosphate oxidised to?
- what is the oxidant in this case?
- Referring to one of the metabolic
maps in the text....
- What is the metabolic importance of chorismic acid (or
"chorismate")?
-
In which of the following organisms does chorismic acid metabolism
actually occur?
- carrots
- rabbits
- mushrooms
- earthworms
- common bacteria
- describe 2 different mechanisms whereby micro-organisms
can take up up ammonia (NH3) from the medium in
whcih they are growing.
- Write short essays/notes on the following topics
- Transcriptional and translational control in different types
of organism
- The importance of the cro gene
to lambdoid phages
TOP of page
Topic 8
- A chef in a large restaurant or hotel decides to sort out
the enormous number of pans that have been accumulated over the
years. Design an Adansonian scheme for helping the chef differentiate
frying pans, sauce pans, woks, griddle pans....
- Without drawing a time scale, sketch a phylogenetic tree for
the following: bumble bee, gold fish, earth worm, star fish, pig,
cat.
-
What is the time scale in question 2?
- For what taxonomic purpose(s) are mtDNA sequences
useful?
- Comment on
the inheritance of mtDNA.
- What macromolecular studies would be useful for studying
relationships between
- conifers
- zoöplankton
- daisies and related plants
- isolates of HIV
- Comment on mistakes which would be made by putting execessive
taxonomic reliance on the toxicity of different species of snake
on the basis of the extent to which they are venemous.
- Use the
link
provided to answer these questions about
enzymes
-
What is EC 2.1.1.1 ?
-
How many EC numbers describe DNA polymerases?
-
What is the EC number for rhodanese?
-
How does the EC cope with "restriction enzymes"?
- Name one other Web site for relating
EC numbers to enzymes.
TOP of page
Topic 9
- In the case of the fictitous (and rather implausible!)
fish how might the frequency of
recombinants between genes 1-9 be related to the spacing between the
genes ?... and how was this used in "classical
genetics"?
- Explain the terms
- Explain the terms
- transposon
- plasmid
- resolution (in relation to transposition)
- Be prepared to discuss the molecular a clinical implications
of drug resistance.
- Name 2 drug resistance mechanisms
used in bacteria of clinical importance
- Name/describe 3 ways in which DNA damage can arise
in vivo... and
- name/describe repair mechanisms for your 3 examples
(question 6).
-
In experiments with experimental micro-organimsms, caffeine is not
in itself mutagenic but it enhances the mutagenicity of mutagens
including UV light. Speculate plausibly on the mechanism for this
effect of caffeine.
-
What sort of chemical is caffeine?
- What is "Darwinism": hypothesis, wild guess,
belief... or what?
- Name 3 factors that might account for a very rapid generation
of species diversity over a relatively short period of time.
TOP of page


